Today we did an in class review to get ready for the test in the form of Jeopardy!
Here's a copy for you to play at home. Try to answer all the questions to help you study. The 1000 point questions are particularly challenging.
PowerPoint: Chemistry Review Jeopardy
Good luck in your studies!
Tuesday, 8 October 2013
Monday, 7 October 2013
Oct. 7 – Work Period
Today in Period 1 we had an anti-bullying assembly. Here is a powerful video on the topic of bullying and how it affects people.
Video: To This Day Project - Shane Koyczan
Here's a live version where you can see the author reciting it in person.
Video: To This Day - Live version
Then in class I handed back the Physical and Chemical Changes Investigation. Have a look at TeachAssist for your updated marks.
Here are some notes on your responses:
Video: To This Day Project - Shane Koyczan
Here's a live version where you can see the author reciting it in person.
Video: To This Day - Live version
_________________________________________________________________
Then in class I handed back the Physical and Chemical Changes Investigation. Have a look at TeachAssist for your updated marks.
Here are some notes on your responses:
- Clarity is either transparent, translucent or opaque.
- Dissolving is spelt D-I-S-S-O-L-V-I-N-G. Breaking of a substance without affecting the particles.
Wrong: the magnesium dissolved.
Correct observation: I can no longer see the magnesium. - Evaporation is a phase change from liquid to gass.
Wrong: the magnesium evaporated.
Correct observation: I can no longer see the magnesium. - Both dissolving and evaporation are physical changes.
- "Cannot reverse" is not an observation.
- You can ONLY use your 5 senses to make observations.
- Use these observations to support "How do you know".
The rest of the period was for working on the element project and for studying.
Here are some review questions to help you study:
- Ch. 5, P. 202 #1, 2, 7, 10, 11, 12, 16, 17, 18, 19
- Ch. 6, P. 248 #1, 2, 4, 5, 6, 7, 10, 11, 13, 14, 19
- Ch. 7, P. 280 #1, 2, 3, 5, 6, 7, 8, 9
Friday, 4 October 2013
Oct. 4 – Naming Chemicals
Here are the solutions to yesterday's homework.
Here we go, the final topic before the test!
Success Criteria: Given the chemical formula for a certain compound, you can write down its chemical name.
Handout: Naming Compounds
There are a lot of rules, so take care in reading the handout carefully.
There will be no new topics between now and the unit test! For those of you anxious to get started on studying, here are the relevant questions to try from each chapter:
Here we go, the final topic before the test!
Naming compounds
Learning Goals: Understand the conventions in naming chemical compounds.Success Criteria: Given the chemical formula for a certain compound, you can write down its chemical name.
Handout: Naming Compounds
There are a lot of rules, so take care in reading the handout carefully.
Element Project – Due Oct. 10
Handout: Element Project
In this project you will become an expert on one of the elements in the periodic table. Use the handout as a guide to what information you should look for. Then, once you've gather all the information, put it together into a pamphlet (or brochure) that describes that element. You can do it by hand or typed, but make sure you print it if you use a computer.
Unit Test – Oct. 9
Handout: Topics to studyThere will be no new topics between now and the unit test! For those of you anxious to get started on studying, here are the relevant questions to try from each chapter:
Ch. 5, P. 202 #1,
2, 7, 10, 11, 12, 16, 17, 18, 19
Ch. 6, P. 248 #1,
2, 4, 5, 6, 7, 10, 11, 13, 14, 19
Ch. 7, P. 280 #1, 2, 3, 5, 6, 7, 8, 9
Thursday, 3 October 2013
Oct. 3 – Ionic Formulas and Covalent Bonds
Learning Goals:
- Understand how ionic bonds form.
- Understand how covalent bonds form.
Success Criteria:
- You can write the formula for ionic compounds.
- You can explain how covalent bonds are formed between two non-metals.
Here's an interactive review of what we worked on yesterday:
Next we discussed the patters on the periodic table and how to figure out the ionic charges of each element.
Handout: Valence
Using these valences, we can figure out how many ions of each element are required to make an ionic compound. I got some volunteers to come up and demonstrate using black and white beads as electrons and protons. Then we discussed how you can use the crisscross method to figure out the same thing.
Handout: Writing formulas for ionic compounds
Finally we talked about covalent bonds. Again, students came up to the front to demonstrate using beads as electrons. In a covalent bond, the atoms don't lose or gain electrons, they share!
If two hydrogen each share their electrons, then they will each have two, making a more stable molecule.
Here's the example I showed in class of two oxygen forming a covalent bond with a carbon.
Homework: Complete the handouts.
Wednesday, 2 October 2013
Unit Test!
The Chemistry Unit Test will be on Wednesday Oct. 9.
We will wrap up the material this week and have a couple days of review before the test. If there's anything in the unit so far that you are not sure about, please make sure to clarify with me before the test.
Handout: Topics to Study
We will wrap up the material this week and have a couple days of review before the test. If there's anything in the unit so far that you are not sure about, please make sure to clarify with me before the test.
Handout: Topics to Study
Oct. 2 – Ionic Bonds
Learning Goals: Understand how ionic bonds are formed.
Success Criteria: You can draw the Bohr-Rutherford diagram for ions and show how they bond.
Today we reviewed som Bohr-Rutherford diagrams and I showed you what Lewis dot diagrams are. These diagrams are only concerned with the outer shell of each atom. Here are some examples:
We spent some time discussing the patterns that we see. All group 1 atoms have one electron in the outer shell. All nobel gases have complete outer shells.
Here are the handouts for today:
Here's the main idea you should get out of these handouts: ionic bonds form when a metal loses one or more electrons and a non-metal gains the lost electrons.
 |
| The Bohr-Rutherford diagrams. |
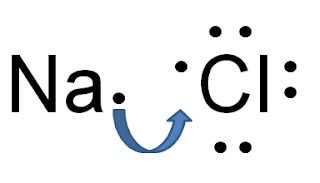 |
| The Lewis dot structures. |
 |
| Positive and negative ions attract each other and join together forming ionic compounds. |
Homework: Complete the handout on Bohr-Rutherford diagrams for ions.
By popular demand, here are the lyrics to the periodic table song!
Video: Periodic Table Song
Written, Directed, Produced, Edited and Sung by Mitchell Moffit.
Based on the "Can-Can" music, by Offenbach.
LYRICS:
There's Hydrogen and Helium
Then Lithium, Beryllium
Boron, Carbon everywhere
Nitrogen all through the air
With Oxygen so you can breathe
And Fluorine for your pretty teeth
Neon to light up the signs
Sodium for salty times
Magnesium, Aluminium, Silicon
Phosphorus, then Sulfur, Chlorine and Argon
Potassium, and Calcium so you'll grow strong
Scandium, Titanium, Vanadium and Chromium and Manganese
CHORUS
This is the Periodic Table
Noble gas is stable
Halogens and Alkali react aggressively
Each period will see new outer shells
While electrons are added moving to the right
Iron is the 26th
Then Cobalt, Nickel coins you get
Copper, Zinc and Gallium
Germanium and Arsenic
Selenium and Bromine film
While Krypton helps light up your room
Rubidium and Strontium then Yttrium, Zirconium
Niobium, Molybdenum, Technetium
Ruthenium, Rhodium, Palladium
Silver-ware then Cadmium and Indium
Tin-cans, Antimony then Tellurium and Iodine and Xenon and then Caesium and...
Barium is 56 and this is where the table splits
Where Lanthanides have just begun
Lanthanum, Cerium and Praseodymium
Neodymium's next too
Promethium, then 62's
Samarium, Europium, Gadolinium and Terbium
Dysprosium, Holmium, Erbium, Thulium
Ytterbium, Lutetium
Hafnium, Tantalum, Tungsten then we're on to
Rhenium, Osmium and Iridium
Platinum, Gold to make you rich till you grow old
Mercury to tell you when it's really cold
Thallium and Lead then Bismuth for your tummy
Polonium, Astatine would not be yummy
Radon, Francium will last a little time
Radium then Actinides at 89
REPEAT CHORUS
Actinium, Thorium, Protactinium
Uranium, Neptunium, Plutonium
Americium, Curium, Berkelium
Californium, Einsteinium, Fermium
Mendelevium, Nobelium, Lawrencium
Rutherfordium, Dubnium, Seaborgium
Bohrium, Hassium then Meitnerium
Darmstadtium, Roentgenium, Copernicium
Ununtrium, Flerovium
Ununpentium, Livermorium
Ununseptium, Ununoctium
And then we're done!!
Tuesday, 1 October 2013
Oct. 1 - Bohr-Rutherford Diagrams
I handed back the Density Quiz today. Here are the solutions:
Next we discussed an interesting element: carbon. Carbon can come in may different forms, each with vastly different properties.
Handout: Graphite to Diamonds
Success Criteria: You can draw the Bohr-Rutherford diagrams for the first 20 atoms on the periodic table.
Next we discussed an interesting element: carbon. Carbon can come in may different forms, each with vastly different properties.
Handout: Graphite to Diamonds
Bohr-Rutherford Diagrams
Learning Goals: Understand the structure of atoms.Success Criteria: You can draw the Bohr-Rutherford diagrams for the first 20 atoms on the periodic table.
First you must know how to find the number of protons, neutrons and electrons in each atom. Here's a worksheet for you to practice:
Handout: Subatomic Particles Worksheet
Next we went over how to draw Bohr-Rutherford diagrams. Here are the steps:
- Determine the number of protons and neutrons.
- Write it in the middle (nucleus.)
- Determine the number of electrons.
- Put the electrons in concentric shells (orbitals)
- First orbital can hold up to 2 electrons.
- Second orbital can hold up to 8 electrons.
- Third orbital can hold up to 8 electrons.
- Fourth orbital can hold up to 8 electrons.
Handout: Bohr-Rutherford Diagrams
Here's an example: http://www.youtube.com/watch?v=wy83UlGQpWw
Here are the solutions:
Homework: Complete the handouts and answer the questions to find patterns with these diagrams.
 |
| Dr. Manhattan from Watchmen has a Bohr-Rutherford diagram of hydrogen on his forehead! |
Subscribe to:
Posts (Atom)








